 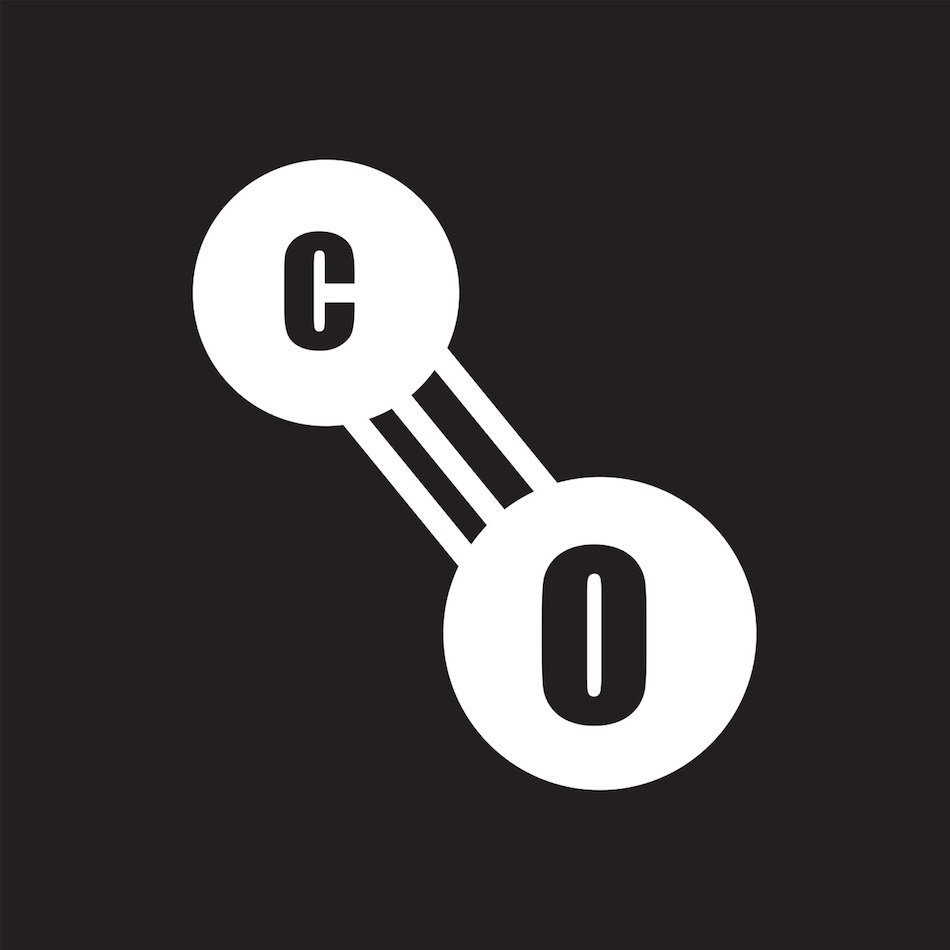 
Both experiment and more advanced models show that the triple-bonded form (a) predominates. Structures that do not obey the octet rule (for carbon):ī) For :C:O::: Carbon: 4 – 2 – 1 = +1 Oxygen: 6 – 6 – 1 = –1Ĭ) For :C::O:: Carbon: 4 – 2 –2 = 0 Oxygen: 6 – 4 – 2 = 0Ĭomment: All three structures are acceptable (because the formal charges add up to zero for this neutral molecule) and contribute to the overall structure of carbon monoxide, although not equally. Write out some structures for carbon monoxide CO, both those that do and do not obey the octet rule, and select the "best" on the basis of the formal charges.Ī) For :C:::O: Carbon: 4 2 3 = –1 Oxygen: 6 2 3 = +1 (Bear in mind that all four positions around the carbon atom are equivalent in this tetrahedral molecule, so there are only four chloromethanes.) The manner in which the individual bonds contribute to the dipole moment of the molecule is nicely illustrated by the series of chloromethanes: In this example for methanol CH 3OH, the blue area centered on hydrogen represents a positive charge, the red area centered where we expect the lone pairs to be located represents a negative charge, while the light green around methyl is approximately neutral. A good example of this is carbon monoxide, in which the partial negative charge resides on the carbon, as predicted by its negative formal charge (below.)Įlectron densities in a molecule (and the dipole moments that unbalanced electron distributions can produce) are now easily calculated by molecular modelling programs. In molecules containing nonbonding electrons or multiple bonds, the elecronegativity difference may not correctly predict the bond polarity. The nonbonding pairs on oxygen are a contributing factor to the high polarity of the water molecule. H 2O, by contrast, has a very large dipole moment which results from the two polar H–O components oriented at an angle of 104.5°. The dipole moment corresponding to an individual bond (or to a diatomic molecule) is given by the product of the quantity of charge displaced q and the bond length r: The magnitude of interaction with the electric field is given by the permanent electric dipole moment of the molecule. Dipoles are conventionally represented as arrows pointing in the direction of the negative end. Such bonds are said to be polar and to possess partial ionic character, and they may confer a polar nature on the molecule as a whole.Ī polar molecule acts as an electric dipole which can interact with electric fields that are created artificially or that arise from nearby ions or polar molecules. As a consequence, the electrons will not be shared equally the center of the negative charges in the molecule will be displaced from the center of positive charge. The gray arrows pointing away from each other represent repulsionīecause each end of a dipole possesses only a fraction of the charge of an electron, dipole–dipole interactions are substantially weaker than the interactions between two ions, each of which has a charge of at least ☑, or between a dipole and an ion, in which one of the species has at least a full positive or negative charge.When non-identical atoms are joined in a covalent bond, the electron pair will be attracted more strongly to the atom that has the higher electronegativity. (CC BY-SA-NC anonymous) The green arrows pointing towards each other represent attraction. \): Both attractive and repulsive dipole–dipole interactions occur in a liquid sample with many molecules.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
